Colouring materials (Dyes/pigments) are generally coloring materials for dyeing operation. There are various types of dyes & pigments for the coloration of textiles.
Properties of Dyes
The four properties that dyes must possess are:
- Intense color
- In almost all cases solubility in on aqueous solution (either permanent or only during the dyeing operation)
- Ability to be absorbed on and retained by the fibre (substantivity) or to be chemically combined with it.
- Fastness i.e., the ability when attached to a fibre to withstand the treatment the fibre undergoes in manufacturing process and in normal use.
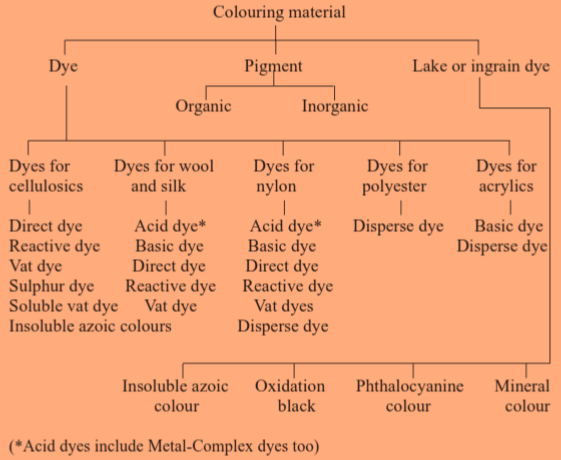
Classification
Coloring materials are classified in several aspects such as based on their origin, chemical structure, field of application, ionic nature, solubility in water etc.
1. Source of origin
a) Natural colors
The word „natural dye‟ covers all the dyes derived from the natural sources like plants, animals and minerals. Natural dyes are mostly non-substantive and must be applied on textiles by the help of mordants, usually a metallic salt, having an affinity for both the coloring matter and the fibre. They are non-toxic, eco-friendly. Some of its constituents are anti-allergens, hence prove safe for skin contact and are mostly non-hazardous to human health.
b) Synthetic colors
A large amount of synthetic colors are obtained through reaction of dye intermediates, e.g.,anthranilic acid, β-napthol, benzidine, naphthionic acid, J-acid, metanilic acid, H-acid, hydroquinone, Schaffer‟s acid, G-acid, R-acid etc., with other chemicals. These intermediates are first synthesized through various chmical reactions and are stored as starting material to produce various dyes.
2. Chemical structure
Coloring materials are also classified based on their chemical structure, e.g., azo, nitro, nitroso, stilbene, di- and tri-phenylmethane, xanthane, acridine, quinolone, methane, thiaole, azine, oxazine, thiazine, anthraquinone, indigoid and pthalocyanine, etc. examples of dyes with these groups will be found separately in different chapters.
3. Field of application
A. Readymade Dyes
i) Water Soluble Dyes
a) Direct dyes
b) Acid dyes
c) Basic dyes
d) Reactive dyes
e) Optical whiteners |
ii) Water Insoluble Dyes
a) Vat dyes
b) Sulphur dyes
c) Disperse dyes |
B. Ingrain Dyes
a) Azoic colors
b) Oxidation colors
c) Mineral colors
a. Dyes are colored compounds which are absorbed or adsorbed by the fibre from a solution or suspension where they are subsequently fixed and are thus used for the coloration of textiles. The term also applies to certain colorless compounds (optical brighteners) and those which only form an insoluble dye through combination on or in the fibre (e.g. napthols and Turkey Red).
b. Pigments do not fit this definition since they are water-insoluble colorants having no affinity for textile fibres and, for this reason, have to be applied with a binder capable of bonding to the fibre. consequently, pigments must be regarded as a special case in textile coloration.
c. Lakes or Ingrain dyes are such pigments that are formed in situ fibre, e.g. phthalocyanine blue formed through reaction between copper complex and alcian blue; insoluble azoic colors formed in situ through reaction of naphthol (coupling component) and a diazotized base; reaction product of metal oxides in situ fibres, etc.
4. Others
(i) Ionic nature
Dyes can also be classified according to electrical charge that is exhibited by its ionized colored part before or during dyeing. Based on this, direct, acid, reactive, sulphur, metalcomplex, vat and soluble vat dyes are anionic; basic dye is cationic and disperse dye is non-ionic in nature.
(ii) Solubility in water
Direct, reactive, solubilized vat and acid dyes are water soluble dyes. Vat and sulphur dyes are water insoluble but can be taken into solution by reduction and solubilisation. Basic dye is soluble in acidic warm bath while other dyes are insoluble in water under normal conditions.
Texpedi.com
Check out these related articles: