Nanoparticles | Green synthesis and applications for Waste Water Treatment: Nowadays, many countries, particularly in the developing world, grapple with drinking water issues. The world confronts significant challenges in meeting the growing demand for clean water as freshwater supplies diminish due to:
- extended droughts,
- population growth,
- more stringent health based regulations and
- competing demands from a variety of users.
Several industries, including paper and pulp, tanneries, cosmetics, and more, discharge toxic effluents into water. Consequently, clean water, devoid of harmful chemicals and pathogens, is crucial for human health. The protection of water treatment systems against potential chemical and biological terrorist acts is also becoming a critical issue in water resources planning.
Water treatment techniques:
Today, we employ diverse techniques to treat water. Additionally, we use chemical and physical agents like chlorine and its derivatives. Above all, ultraviolet light, boiling, and low-frequency ultrasonic irradiation are common methods. Afterward, distillation and reverse osmosis become effective. If we need more options, we can turn to water sediment filters (fiber and ceramic), activated carbon, solid block, pitcher, and faucet-mount filters, as well as bottled water. All in all, ion exchange water softeners, ozonating, and activated alumina “altered” water provide further choices.
People widely use halogens like chlorine (Cl) and bromine (Br) as antibacterial agents. However, their direct use faces issues due to high toxicity and vapor pressure in pure form. The most common cation in water affecting human and animal health is NH4+.
Ammonia removal is vital in drinking water. It prevents oxygen depletion and algae blooms, as it’s highly toxic to fish. You can replace it with biologically acceptable cations, such as Na+, K+, or Ca2+, in the zeolite. In recent decades, research explored synthetic and natural zeolites, polymer films, and metal ions (Ag+, Cu2+, Zn2+, Hg2+, Ti3+, Ni2+, Co2+) as water disinfectants.
Nanotechnology in water treatment:
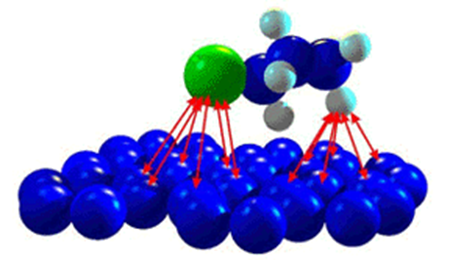
Advances in nanoscale science and engineering suggest that many of the current problems involving water quality could be resolved or greatly diminished by using nonabsorbent, nano catalysts, bioactive nanoparticles, nanostructured catalytic membranes, submicron, nano powder, nanotubes, magnetic nanoparticles, granules, flake, high surface area metal particle supramolecular assemblies with characteristic length scales of 9-10 nm including clusters, micro molecules, nanoparticles and colloids have a significant impact on water quality in natural environment.
Nanotechnology detects pesticides, metals (like Cadmium, copper, lead, mercury, nickel, zinc), nutrients (e.g., Phosphate, ammonia, nitrate, nitrite), and more. It also detects cyanide, algae toxins, viruses, bacteria, parasites, antibiotics, and biological agents related to terrorism.
Innovations in the development of novel technologies to desalinate water are among the most exciting and seem to have promise. Opportunities and challenges of using nanomaterials in the purification of surface water, groundwater and industrial wastewater streams is a matter of continuing concern.
Many people hold misconceptions about nanotechnology. They believe nanoparticles can kill organisms, repair tissue, improve water quality, and cure disease. Recent applications of nanoparticulate silver have included open wound and burn treatment and preliminary studies have shown that a 20-ppm silver colloidal suspension (~30 nm diameter) in purified water has a 100% cure rate for malaria.
Titanium dioxide, especially as nanoparticulate anatase, is also an interesting antibacterial, with notable photocatalytic behavior. But ultrafine anatase has also been identified as cytotoxic and in-vivo studies have shown that it can be severely toxic in the respiratory system. Nano capsules and nanodevices may present new possibilities for drug delivery, gene therapy, medical diagnostics, antimicrobial activity etc.
Laboratory-scale experiments investigate the effect of particle size on heavy metal adsorption by iron oxide and titanium dioxide nanoparticles. These nanoparticles are excellent sorbents for metal contaminants. Furthermore, spherical aggregates of nanoparticles closely resemble the resin beads used in water purification.
Moreover, ligands, fluvic acids, hemic acids, and their aggregates significantly impact contaminant mobility, reactivity, and bioavailability. In addition to this, nanoparticles can be tailored for use as separation or reaction media for pollutants.
Not only do nanoparticles possess larger surface areas compared to bulk particles on a mass basis, but they can also be functionalized with various chemical groups to enhance their affinity for target compounds.
Advantages of Nanoparticles:
Researchers are increasingly testing nanoparticles as potential alternatives to traditional adsorbents. Moreover, they choose them because nanoparticles have a vast surface area, perfect mechanical rigidity, adjustable surface chemistry, and can be feasibly regenerated under mild conditions. Additionally, nanoparticles offer additional advantages over other adsorbent materials. Not only can manufacturers easily produce them with a wide range of physicochemical properties, but also they are highly effective.
Recovering and recycling nanoparticles is especially difficult because they tend to form complex, hard-to-separate mixtures with other substances. It is of prime importance to design a new process which is perfectly suited to recycling nanoparticle.
Check out these related articles: