Viscose Rayon Fibre is often referred to as semi-synthetic fibre. It is produced from naturally originated cellulose in the laboratory conditions. That is why it is also called regenerated cellulose fibre. The chemical reactions that are involved in the manufacture of viscose are as follows:
Viscose rayon manufacturing:
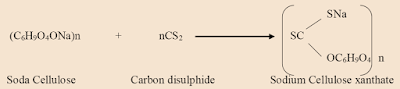
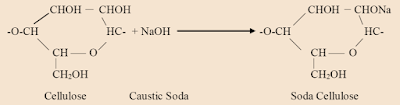
The cellulose is treated with a 17.5% solution of caustic soda (NaOH) which converts it into soda cellulose.
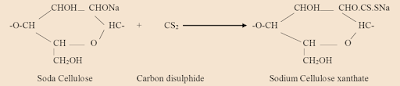
The reaction between the soda cellulose and the carbon disulphide may be represented:
Each glucose residue in the cellulose polymer chain must be thought of as reacting in the following way:
The reaction takes place on the hydroxyl group (OH-) in position 2 in the glucose residues in the cellulose polymer. When the sodium cellulose xanthate is dissolved in weak caustic soda to form the viscose solution known as “VISCOSE” the xanthate radicals probably combine loosely with the molecules of caustic soda.
When the viscose solution is first made, it is very thick. On standing, it becomes thinner and then, later, more viscose again. This process is known as “Ripening”.
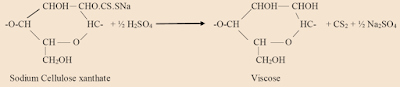
Now, Sodium Cellulose xanthate is spun into a coagulating bath of sulphuric acid, H2SO4 which completes the conversion of the Sodium Cellulose xanthate to cellulose.
This complete the series of chemical reactions by which viscose is made. Essentially they consist of the following stages:
1. Wood cellulose and concentrated caustic soda react to form soda cellulose.
2. The soda cellulose reacts with carbon disulphide to form sodium cellulose xanthate.
3. The sodium cellulose xanthate is dissolved in dilute caustic soda to give a viscose solution.
4. The solution is ripened.
5. It is extruded into sulphuric acid which regenerates the cellulose, now in the form of long filaments (Viscose Rayon).
Reference Book
- MAN-MADE FIBRES
- R. W. MONCRIEFF
Emdadul Haq
Lecturer, Department of Textile Engineering, Primeasia University; Bangladesh
Texpedi.com
Check out these related articles:







kindly visit here https://pokerbitcoin.info
Cash is the lifeblood of a business. No business owner wants to see their business struggling because of lack of funds or in debt due to mismanaged finances. What they want to see is a regular flow of profit but this is easier said than done. a course in miracles blog
I rattling pleased to find this internet site on bing, just what I was searching for also saved to my bookmarks. cse.google.com.ec/url?sa=t&url=https%3A%2F%2Fwijidigital.com%2F
Great web site. Plenty of useful information here. I am sending it to several buddies ans also sharing in delicious. And naturally, thanks for your sweat! Authority Backlinks
I likewise believe thus, perfectly pent post! . kasthooriman serial cast
I likewise believe thus, perfectly pent post! . kiss918
But a smiling visitor here to share the love (:, btw great pattern . https://totoya24.com
emember that the composition is an argument: the job in an essay isn't cheapest essay writing service
you are talking about the composite materials fabrication method. Anyway, thanks for being with us.
Pressure is provided by vacuum bags. Compaction pressure consolidates the plies. Release agents applied to the tool may be liquid release coatings, release waxes, or barrier tape. carbon fibre mold release agent
Thanks Texpedia for publishing the article.
BOSS